Cryogenic Heat Transfer
Cryogenic heat transfer is the process by which energy in the form of heat is exchanged between bodies at different temperatures. Heat is generally transferred through convection, radiation, or conduction.
- Convection
- Conduction
- Radiation
- Conduction Calculations
- Radiation Calculations
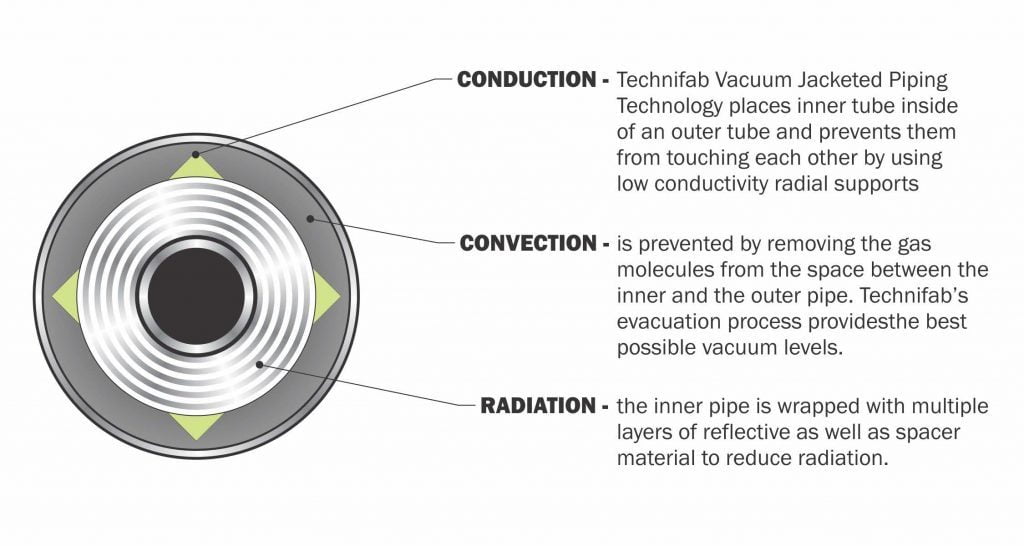
Convection
Convection may occur within a body or between two bodies if they are brought into contact. The fluid motion will almost certainly occur if one of the bodies is a liquid or gas. Convection is the conduction process between a solid surface and a moving fluid. The motion of the fluid may be natural or forced. Convection cannot occur in a vacuum. Convection above a hot surface occurs because hot air expands, becomes less dense, and rises. Hot water is less dense than cold water and rises, causing convection currents, which transport energy. Convection can lead to circulation in a liquid, as in heating a pot of water over a flame. Heated water expands and becomes more buoyant. Cooler, denser water near the surface descends, and patterns of circulation are formed.
Conduction
Conduction is the only method of heat transfer in opaque solids. If the temperature at one end of a metal rod is raised by heating, then heat is conducted to the colder end raising its temperature. Conduction is heat transfer utilizing molecular agitation within a material without any motion of the material. Suppose one end of a metal rod is at a higher temperature. In that case, energy will be transferred down the rod toward the colder end because higher-speed particles will collide with the slower ones, with a net transfer of energy to the slower ones.
Radiation
Radiation fundamentally differs from conduction and convection in that the bodies exchanging heat are not in contact. They are many times separated by a vacuum space. All substances emit radiant energy merely by having a positive absolute temperature. The higher the temperature, the higher the amount of energy radiated. All substances also are capable of absorbing radiated energy. Generally, dull, rough surfaces absorb more heat than bright polished surfaces. Bright surfaces reflect more energy than dull surfaces. It is generally accepted that good emitters are sound absorbers, while poor emitters are poor absorbers.
General Heat Conduction Equation
Consider that a rectangular body of length “l” and opposite faces of area “A”, has one face at temperature t’ and the other face at temperature t. Then (t’-t)/l is the temperature drop per unit distance called the temperature gradient. The quantity of heat transmitted per second (Q) from one face to the opposite face is proportional to the area of the face and the temperature gradient.
The proportionality constant “k” depends upon the nature of the substance conducting the heat. The value “k” is the substance’s thermal conductivity (coefficient of thermal conductivity).
It logically follows that “k” for a given substance is the amount of heat conducted per unit time per unit area per unit temperature gradient.
When Q is expressed in terms of BTU/Hour, area in square feet, (t’-t”) in degrees F, and “l” in feet, then k must be expressed in terms of BTU / Hour /degree F / foot.
In engineering work, the area is expressed in square feet, the temperature gradient in degrees F per inch, and the rate of heat conduction in BTU / Hour. Using these units, k for a given substance is the number of BTU’s conducted per hour per square foot for a temperature gradient of 1 degree F per inch of thickness.
Q = k * area * temperature gradient = kA ((t’-t”)/l)
| Thermal conductivity @ 20°C (k)BTU/h/sq. ft. °F / in | Density @ 20°CKg/m3 | Volumetric heat capacity @ 20°C106 J/m3 | Thermal diffusivity @ 20°C10-8 m2/s | |
|---|---|---|---|---|
| Air | 0.1734 | 1.29 | 0.001 | 1938 |
| Glycerol | 2.011 | 1260 | 3.073 | 9 |
| Water | 4.16 | 1000 | 4.180 | 14 |
| Ice | 14.56 | 917 | 2.017 | 104 |
| Olive oil | 1.18 | 920 | 1.650 | 10 |
| Gasoline | 1.04 | 720 | 2.100 | 7 |
| Methanol | 1.456 | 790 | 2.500 | 8 |
| Silicone oil | 0.694 | 760 | 1.370 | 7 |
| Alcohol | 1.179 | 800 | 2.430 | 7 |
| G-10 Fiberglass | 2.03 | |||
| Aluminum | 1644 | 2700 | 2.376 | 9975 |
| Copper | 2705 | 8960 | 3.494 | 11161 |
| Stainless Steel | 111 | 7900 | 3.950 | 405 |
| Aluminum Oxide | 208 | 3900 | 3.413 | 879 |
| Quartz | 20.80 | 2600 | 2.130 | 141 |
| Concrete | 8.87 | 2200 | 1.940 | 66 |
| Marble | 20.8 | 2700 | 2.376 | 126 |
| Glass | 6.45 | 2600 | 2.184 | 43 |
| Pyrex 7740 | 6.970 | 2230 | 1.681 | 60 |
| PVC | 1.11 | 1300 | 1.950 | 8 |
| PTFE | 1.733 | 2200 | 2.200 | 11 |
| Nylon 6 | 1.734 | 1140 | 1.938 | 13 |
| Corian (ceramic filled) | 7.35 | 1800 | 2.307 | 46 |
| Sand (dry) | 2.43 | 1600 | 1.270 | 28 |
| Sand (saturated) | 18.72 | 2100 | 2.640 | 102 |
| Glass pearls (dry) | 1.25 | 1800 | 1.140 | 16 |
| Glass pearls (saturated) | 5.27 | 2100 | 2.710 | 28 |
| Wood | 2.77 | 780 | 0.187 | 214 |
| Cotton | 0.20 | — | 0.001 | — |
| Leather | 0.97 | — | 0.001 | 59 |
| Cork | 0.49 | 200 | 0.047 | 150 |
| Foam glass | 0.312 | 120 | 0.092 | 49 |
| Mineral insulation materials | 0.28 | 100 | 0.090 | 44 |
| Plastic insulation materials | 0.21 | 50 | 0.100 | 30 |
General Radiation Heat Transfer Equation
The Stefan-Boltzmann equation relates the total amount of radiation emitted by an object to its temperature:
P = e * s * A *(Th4-Tc4)
P = power radiated in watts / square meter / second
e = emissivity
s = 5.6703 * 10-8 watt /( meter2K4)(Stefan-Boltzmann Constant)
A = radiating area in square meters
Th= Temperature of warm surface in K
Tc= Temperature of cold surface in K
Emissivity is the ratio of radiation emitted by a blackbody or a surface and the theoretical radiation predicted. A material’s surface emissivity is a measure of the energy emitted when a surface is directly viewed. Surface emissivity is generally measured indirectly by assuming that e = 1 reflectivity. A single energy bounce is measured and the reflected energy measured.
Material Emmissivity Chart
| Type of Material | Specific Material | Emissivity | |
|---|---|---|---|
| Black Coatings & Materials | Anodize Black | 0.88 | |
| Carbon | Filament | 0.53 | |
| Rough Plate | 0.77 | ||
| Lampblack | 0.84 | ||
| Carbon Black Paint NS-7 | 0.88 | ||
| Catalac Black Paint | 0.88 | ||
| Chemglaze Black Paint Z3O6 | 0.91 | ||
| Delrin Black Plastic | 0.87 | ||
| Ebanol C Black | 0.73 | ||
| Ebanol C Black-384 ESH* UV | 0.75 | ||
| EOI Mid-Temperature Black Coating (Up to 200°C) | 0.965 ±0.005 | ||
| EOI High-Temperature Black Coating (Up to 1400°C) | 0.93 ±0.02 | ||
| GSFC Black Silicate MS-94 | 0.89 | ||
| GSFC Black Paint 3l3-1 | 0.86 | ||
| Hughson Black Paint H322 | 0.86 | ||
| Hughson Black Paint L-300 | 0.84 | ||
| Martin Black Paint N-15O-1 | 0.94 | ||
| Martin Black Velvet Paint | 0.94 | ||
| 3M Black Velvet Paint | 0.91 | ||
| Paladin Black Lacquer | 0.75 | ||
| Parsons Black Paint | 0.91 | ||
| Polyethylene Black Plastic | 0.92 | ||
| Pyramil Black on Beryllium Copper | 0.72 | ||
| Tedlar Black Plastic | 0.90 | ||
| Velesat Black Plastic | 0.85 | ||
| White Coatings & Materials | Barium Sulphate with Polyvinyl Alcohol | 0.88 | |
| Biphenyl-White Solid | 0.86 | ||
| Catalac White Paint | 0.90 | ||
| Dupont Lucite Acrylic Lacquer | 0.90 | ||
| Dow Corning White Paint DC-007 | 0.88 | ||
| GSFC White Paint NS43-C | 0.92 | ||
| GSFC White Paint NS44-B | 0.91 | ||
| GSFC White Paint NS-74 | 0.92 | ||
| GSFC White Paint NS-37 | 0.91 | ||
| Hughson White Paint A-276 | 0.88 | ||
| Hughson White Paint A-276+lO36 ESH UV | 0.88 | ||
| Hughson White Paint V-200 | 0.89 | ||
| Hughson White Paint Z-202 | 0.87 | ||
| Hughson White Paint Z-202+1OOO ESH UV | 0.87 | ||
| Hughson White Paint Z-255 | 0.89 | ||
| Mautz White House Paint | 0.90 | ||
| 3M-401 White Paint | 0.91 | ||
| Magnesium Oxide White Paint | 0.90 | ||
| Magnesium Oxide Aluminum Oxide Paint | 0.92 | ||
| Opal Glass | 0.87 | ||
| OSO-H White Paint 63W | 0.83 | ||
| P764-lA White Paint | 0.92 | ||
| Potassium Fluorotitanate White Paint | 0.88 | ||
| Sherwin Williams White Paint (A8W11) | 0.87 | ||
| Sherwin Williams White Paint (F8WJ2O3O) | 0.82 | ||
| Sherwin Williams F8W2030 w Polasol V6V241 | 0.87 | ||
| Sperex White Paint | 0.85 | ||
| Tedlar White Plastic | 0.87 | ||
| Titanium Oxide White Paint with Methyl Silicone | 0.90 | ||
| Titanium Oxide White Paint with Potassium Silicate | 0.92 | ||
| Zerlauts S-13G White Paint | 0.90 | ||
| Zerlauts Z-93 White Paint | 0.92 | ||
| Zinc Orthotitanate with Potassium Silicate | 0.92 | ||
| Zinc Oxide with Sodium Silicate | 0.92 | ||
| Zirconium Oxide with 650 Glass Resin | 0.88 | ||
| Conductive Paints | Brilliant Aluminum Paint | 0.31 | |
| Epoxy Aluminum Paint | 0.81 | ||
| Finch Aluminum Paint 643-1-1 | 0.23 | ||
| Leafing Aluminum in Epon 828 | 0.36 | ||
| Leafing Aluminum (80-U) | 0.32 | ||
| NRL Leafing Aluminum Paint | 0.24 | ||
| NRL Leafing Aluminum Paint | 0.29 | ||
| Silicone Aluminum Paint | 0.30 | ||
| Dupont Silver Paint 48l7 | 0.49 | ||
| Chromeric Silver Paint 586 | 0.30 | ||
| GSFC Yellow NS-43-G | 0.90 | ||
| GSFC Green NS-53-B | 0.87 | ||
| GSFC Green NS-43-E | 0.89 | ||
| GSFC White NS-43-C | 0.92 | ||
| GSFC Green NS-55-F | 0.91 | ||
| GSFC Green NS-79 | 0.91 | ||
| Anodized Aluminum | Black | 0.82 | |
| Black (2nd Sample) | 0.86 | ||
| Blue | 0.87 | ||
| Blue (2nd Sample) | 0.82 | ||
| Brown | 0.86 | ||
| Chromic | 0.56 | ||
| Clear | 0.76 | ||
| Clear (2nd Sample) | 0.84 | ||
| Green | 0.88 | ||
| Gold | 0.82 | ||
| Plain | 0.04 | ||
| Red | 0.88 | ||
| Sulphuric | 0.87 | ||
| Yellow | 0.87 | ||
| Blue Anodized Titanium Foil | 0.13 | ||
| Metals and Conversion Coatings | Aluminum | Highly Polished | 0.039-0.057 |
| Commercial Sheet | 0.09 | ||
| Heavily Oxidized | 0.20-0.31 | ||
| Surface Roofing | 0.216 | ||
| Alzac A-2 | 0.73 | ||
| Black Chrome | 0.62 | ||
| Black Copper | 0.63 | ||
| Black Irridite | 0.17 | ||
| Black Nickel | 0.66 | ||
| Brass | Highly Polished | 0.028-0.037 | |
| Dull Plate | 0.22 | ||
| Buffed Aluminum | 0.03 | ||
| Buffed Copper | 0.03 | ||
| Constantan-Metal Strip | 0.09 | ||
| Copper | Polished | 0.023 | |
| Thick Oxide Layer | 0.78 | ||
| Dow 23 on Magnesium | 0.67 | ||
| Ebanol C Black | 0.77 | ||
| Electroplated Gold | 0.03 | ||
| Electroless Nickel | 0.07 | ||
| Iron, Polished | 0.14-0.38 | ||
| Cast Iron | 0.60-0.70 | ||
| Mild Steel | 0.20-0.32 | ||
| Iron Plate, Rusted Red | 0.61 | ||
| Sheet Sheet, Rough Oxide Layer | 0.81 | ||
| Gold – Pure, Highly Polished | 0.018-0.035 | ||
| Irridite Aluminum | 0.11 | ||
| Inconel X Foil (1 mil) | 0.10 | ||
| Iron and Steel (Not Stainless) | Kannigen-Nickel Alloy | 0.08 | |
| Steel, Polished | 0.066 | ||
| Lead | Unoxidized | 0.057-0.075 | |
| Gray Oxidize | 0.28 | ||
| Molybdenum | Filament | 0.096-0.202 | |
| Massive, Polished | 0.071 | ||
| Nickel | Polished | 0.072 | |
| Nickel Oxide | 0.59-0.86 | ||
| Plain Beryllium Copper | 0.03 | ||
| Platinum Foil | 0.04 | ||
| Quartz, Rough, Fused | 0.93 | ||
| Stainless Steel | Polished | 0.11 | |
| Machined | 0.14 | ||
| Sandblasted | 0.38 | ||
| Machine Rolled | 0.11 | ||
| Boom-Polished | 0.10 | ||
| Silver – Pure, Polished | 0.0.020-0.032 | ||
| Tantalum Foil | 0.05 | ||
| Tungsten Polished | 0.03 | ||
| Vapor Deposited Coatings | Aluminum | 0.02 | |
| Aluminum on Fiberglass | 0.07 | ||
| Aluminum on Stainless Steel | 0.02 | ||
| Chromium | 0.17 | ||
| Chromiumlon 5-mil Kapton | 0.24 | ||
| Germanium | 0.09 | ||
| Gold | 0.02 | ||
| Iron Oxide | 0.56 | ||
| Molybdenum | 0.21 | ||
| Nickel | 0.04 | ||
| Rhodium | 0.03 | ||
| Silver | 0.02 | ||
| Titanium | 0.12 | ||
| Tungsten | 0.27 | ||
| Misc | Asbestos, Board | 0.96 | |
| Aluminum Oxide (Al2o3)-(12/4) on Buffed Alum Initial | 0.23 | ||
| Aluminum Oxide (Al2O3)(12/4) on Fused Silica | 0.24 | ||
| Brick | Red, Rough, No Gross Irregularities | 0.93 | |
| Fireclay | 0.75 | ||
| Concrete Tiles | 0.63 | ||
| Glass | Smooth | 0.94 | |
| Pyrex, Lead, and Soda | 0.95 | ||
| Porcelain, Glazed | 0.92 | ||
| GSFC Dark Mirror Coating – SiO-Cr-Al | 0.04 | ||
| GSFC Composite SiOx – Al2O3-Ag | 0.68 | ||
| Inconel with Teflon Over coating -1 mil | 0.46 | ||
| Kapton Over coating | 0.57 | ||
| Parylene C Over coating | 0.34 | ||
| Roofing Paper | 0.91 | ||
| Silver Beryllium Copper Coating (AgBeCu) | 0.03 | ||
| Teflon Over coating | 0.38 | ||
| Vespel Polyamide SP1 | 0.90 | ||
| Water | 0.95 | ||
| Tapes & Film | Aclar Film (Aluminum Backing) | 1 mil | 0.45 |
| 2 mil | 0.62 | ||
| 5 mil | 0.73 | ||
| Copper Foil Tape | Plain | 0.02 | |
| Sanded | 0.04 | ||
| Tarnished | 0.04 | ||
| Kapton Film (Aluminum Backing) | 0.08 mil | 0.24 | |
| 0.15 mil | 0.34 | ||
| 0.25 mil | 0.45 | ||
| 0.50 mil | 0.55 | ||
| 1.0 mil | 0.67 | ||
| 1.5 mil | 0.71 | ||
| 2.0 mil | 0.75 | ||
| 3.0 mil | 0.82 | ||
| 5.0 mil | 0.86 | ||
| Kapton Film (Chromium-Silicon Oxide-Aluminum Backing (Green)) 1.0 mil | 0.78 | ||
| 235-3M Black | 0.90 | ||
| 425-3M Aluminum Foil | 0.03 | ||
| 850-3M Mylar-Aluminum Backing | 0.59 | ||
| 7361-Mystic Alummized Kapton | 0.03 | ||
| 7452-Mystic Aluminum Foil | 0.03 | ||
| 7800-Mystic Aluminum Foil | 0.03 | ||
| Y9360-3M Aluminized Mylar | 0.03 | ||

